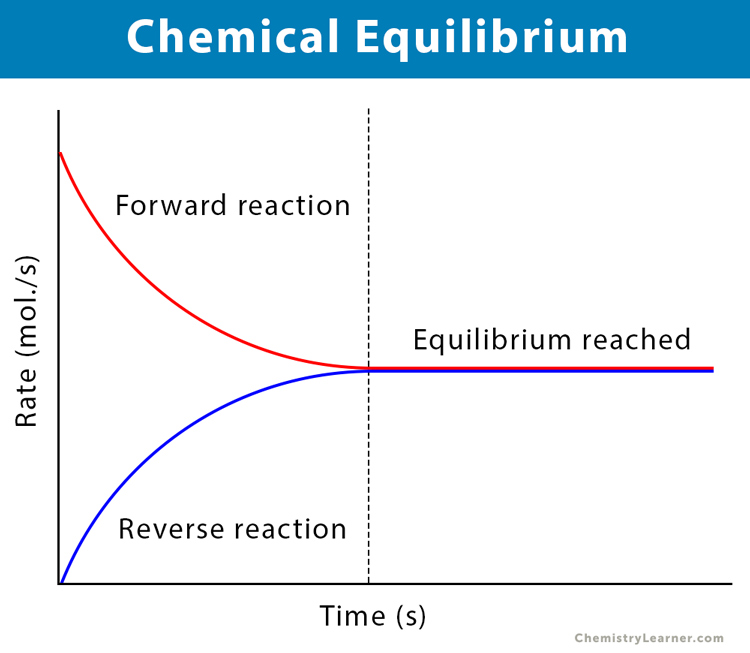
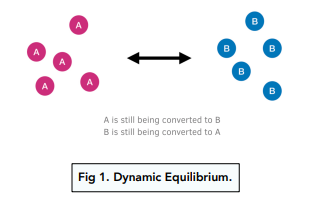
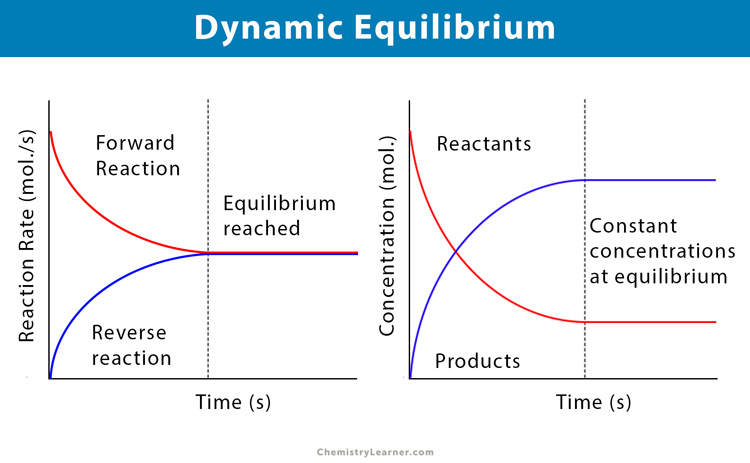
Chemistry Revision Session 7. Equilibria Define the term dynamic equilibrium (3) Consider N 2(g) + 3H 2(g) Fe 2NH 3 (g) ΔH=-100 kJ/Mol Explain. - ppt download

SOLVED:Explain dynamic equilibrium with respect to solution formation. What is a saturated solution? An unsaturated solution? A supersaturated solution?

Chemical Equilibria: Reversible Reactions, Dynamic Equilibrium (1.7.1) | CIE AS Chemistry Revision Notes 2019 | Save My Exams