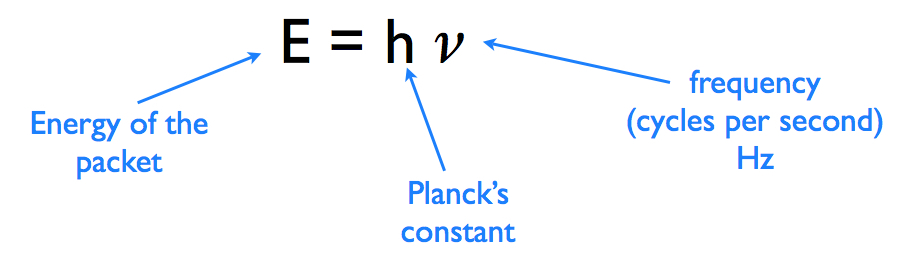
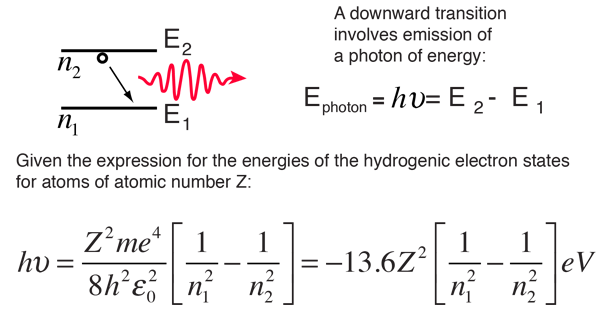
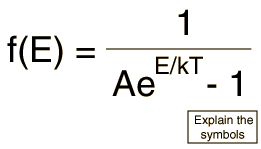Find the ratio of energies of photons produced due to transition of an electron of hydrogen atom from its(i) second permitted energy level to the first permitted level.(ii) the highest permittted energy

OpenStax College Physics Solution, Chapter 30, Problem 25 (Problems & Exercises) | OpenStax College Physics Answers