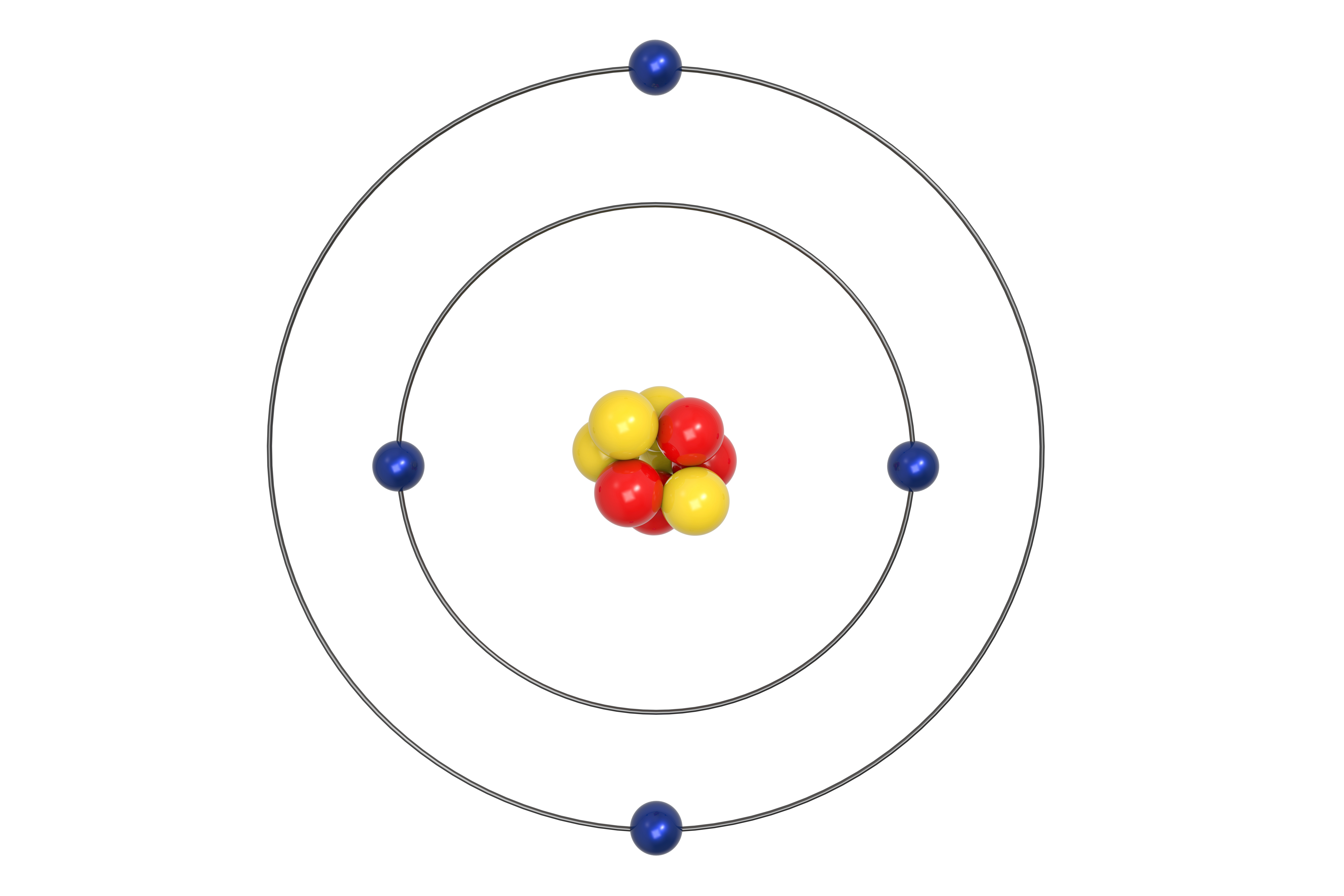
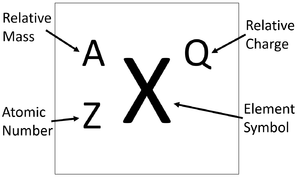
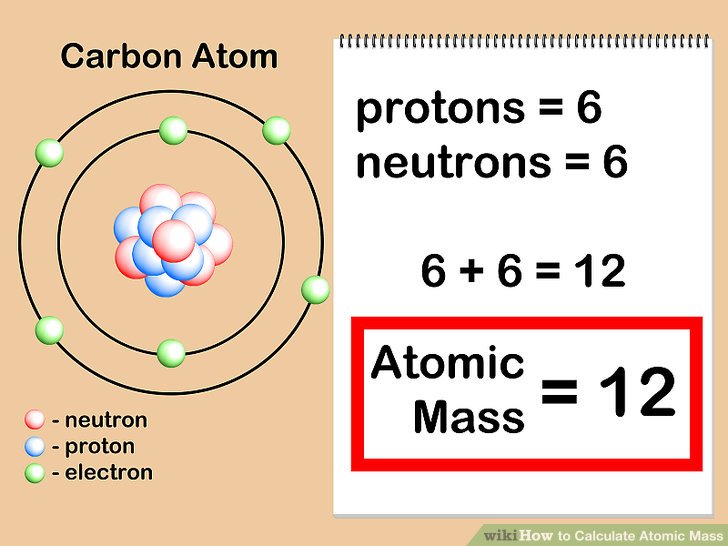
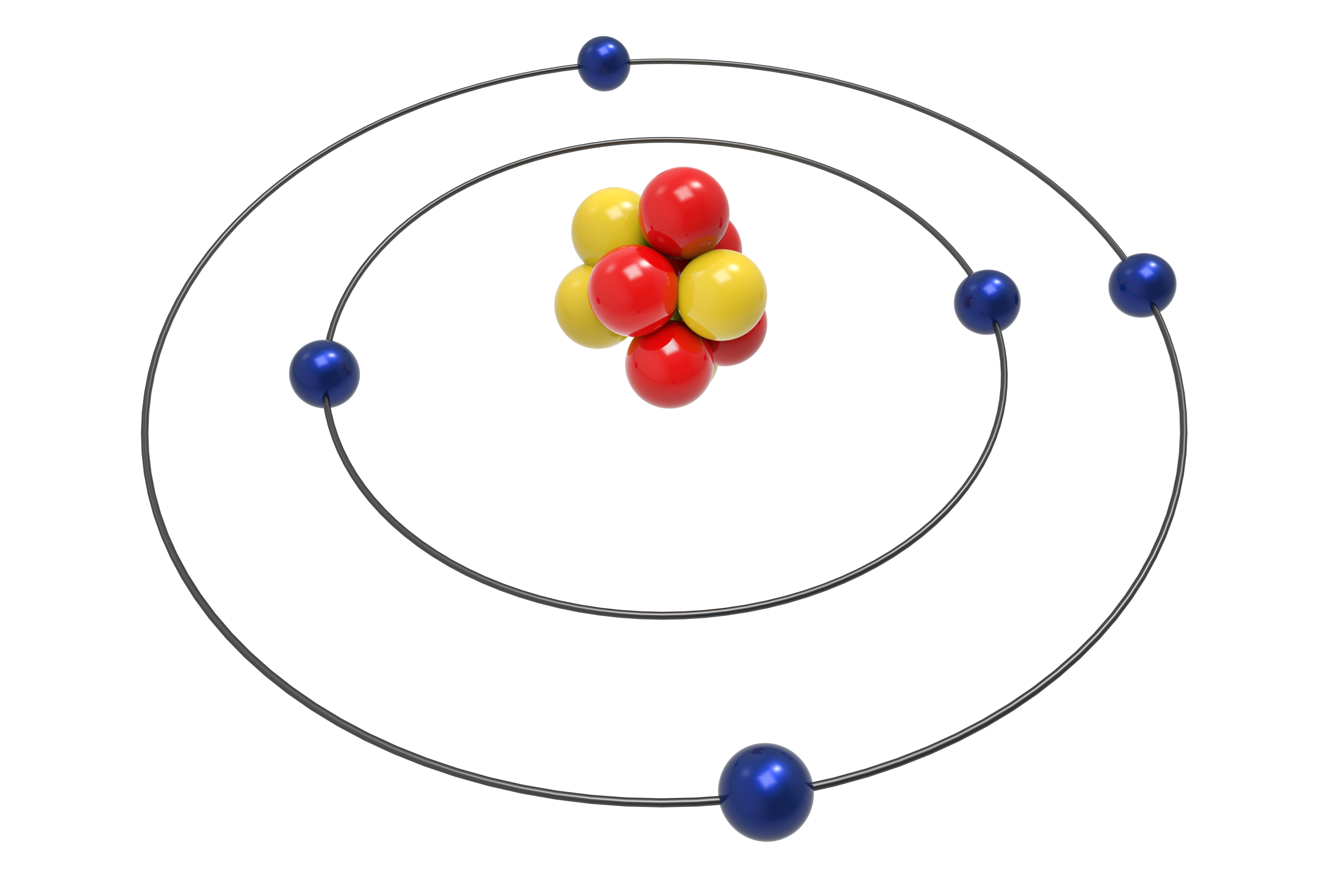
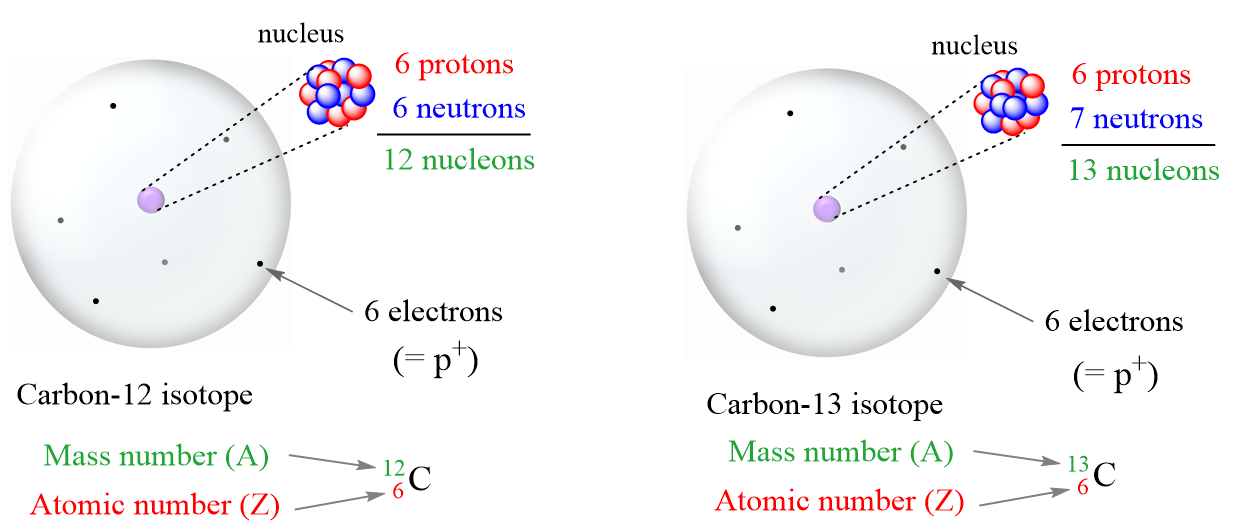
Protons Neutrons Electrons Isotopes - Average Mass Number & Atomic Structure - Atoms vs Ions - YouTube
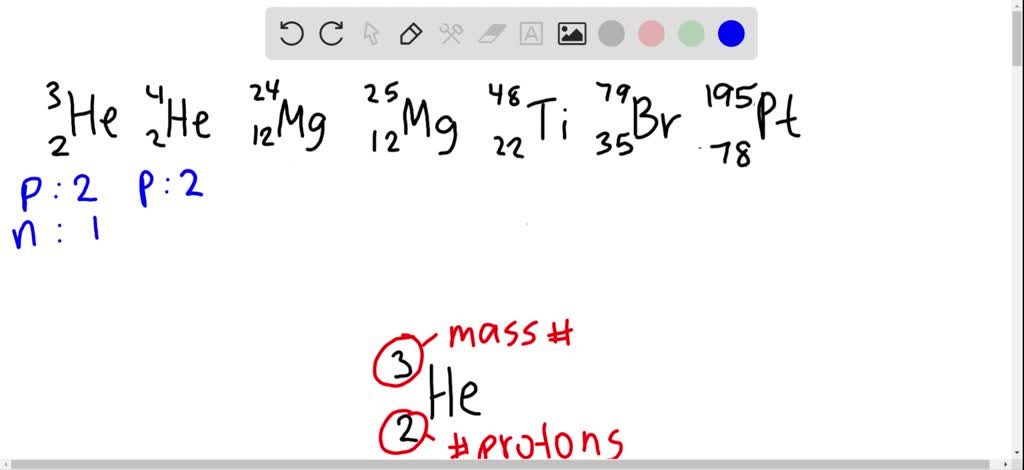
SOLVED:For each of the following species, determine the number of protons and the number of neutrons in the nucleus: 2^3 He,2^4 He,12^24 Mg,12^25 Mg,22^48 Ti,35^79 Br,78^195 Pt
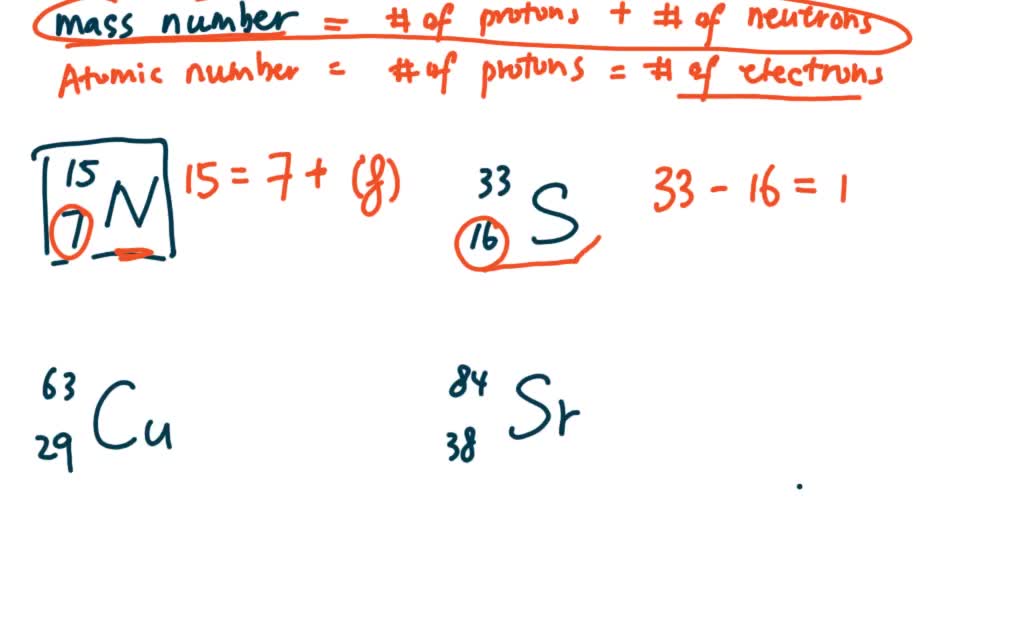
SOLVED:Indicate the number of protons, neutrons, and electrons in each of the following species: 7^15 N,16^33 S,29^63 Cu,38^84 Sr,56^130 Ba,74^186 W,80^202 Hg
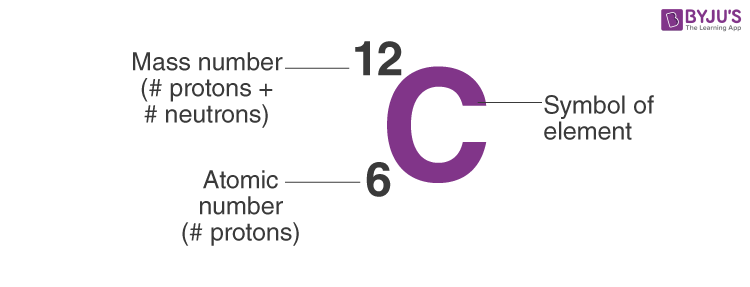
Carbon Protons - What are Protons, Number of protons in Carbon atom, and Uses of Carbon atom along with some FAQs
Calculate the number of electrons,protons,neutrons in 1.phosphorus atom. 2. Phosphate ion. MASS NO.=P=31, O=16 ATOMIC NO.P=15,O=8 along with explanation.