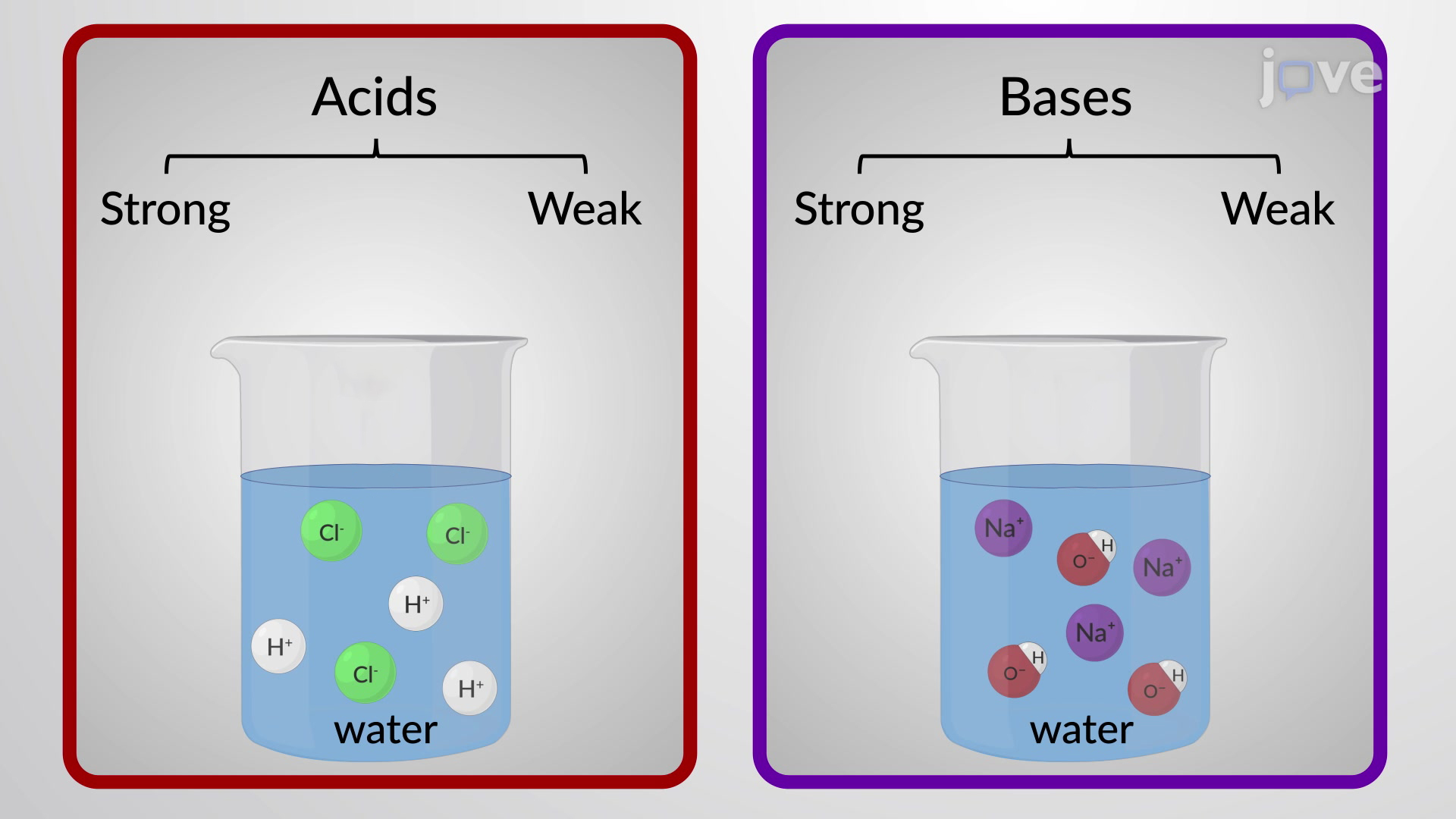
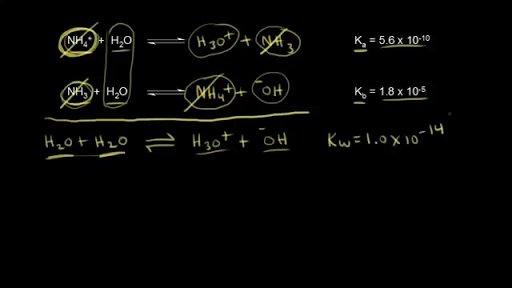SOLVED: The larger Ka (smaller pKa), the smaller Kb (larger pKt): Stronger the acid, weaker its conjugate base, and vice versa Calculate Kb for the base F- anion if Ka = 6.8
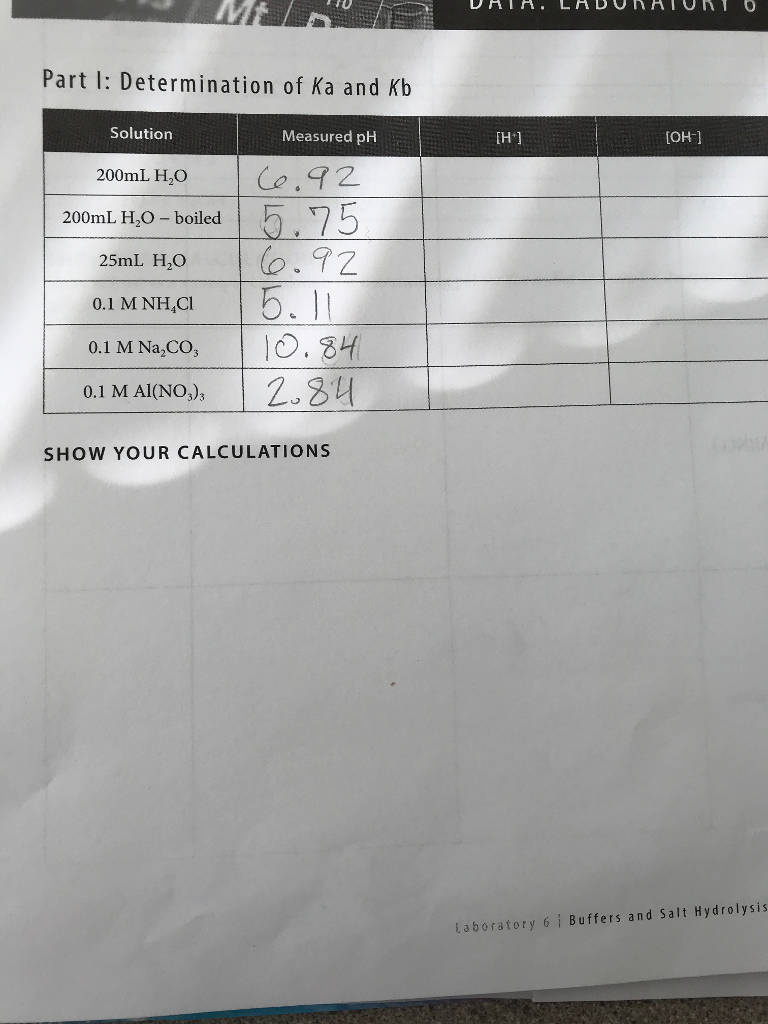
What is value of Ka and kb for water (H20) and how numerically prove that ka ×kb=kw in case of water? - Quora

The acid dissociation constant ka for an unknown acid ha is 4.57 x 10^-3 what is the base dissociation - Brainly.com