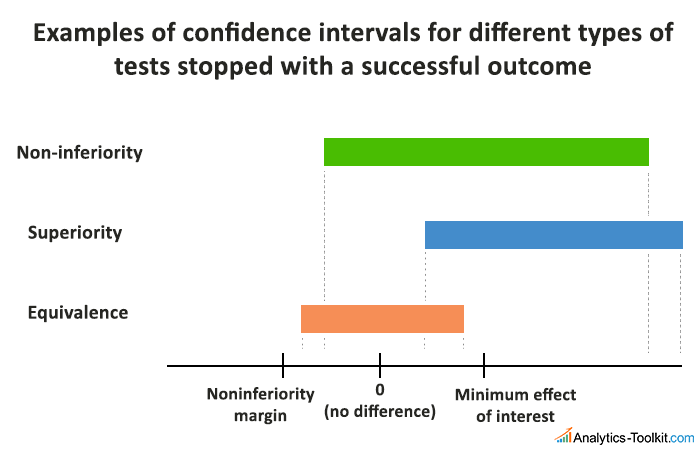
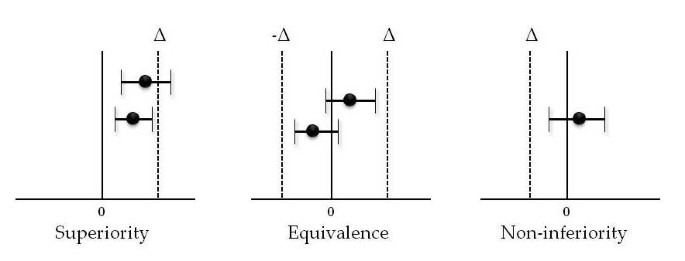
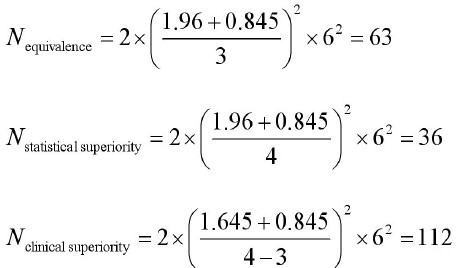Determining the sample size required to establish whether a medical device is non-inferior to an external benchmark | BMJ Open

Sample Size for Non-inferiority Designs|Design Considerations, Dealing with Non-continuous Endpoints - YouTube

Practical guide to sample size calculations: non‐inferiority and equivalence trials - Flight - 2016 - Pharmaceutical Statistics - Wiley Online Library

Pitfalls in reporting sample size calculation in randomized controlled trials published in leading anaesthesia journals: a systematic review - British Journal of Anaesthesia

Differences in sample size for a non-inferiority trial when calculated... | Download Scientific Diagram

Determining the sample size required to establish whether a medical device is non-inferior to an external benchmark | BMJ Open
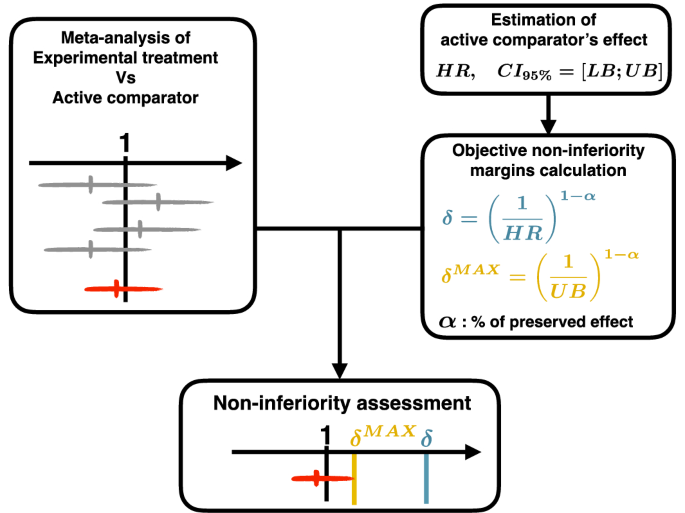
Assessment of non-inferiority with meta-analysis: example of hypofractionated radiation therapy in breast and prostate cancer | Scientific Reports