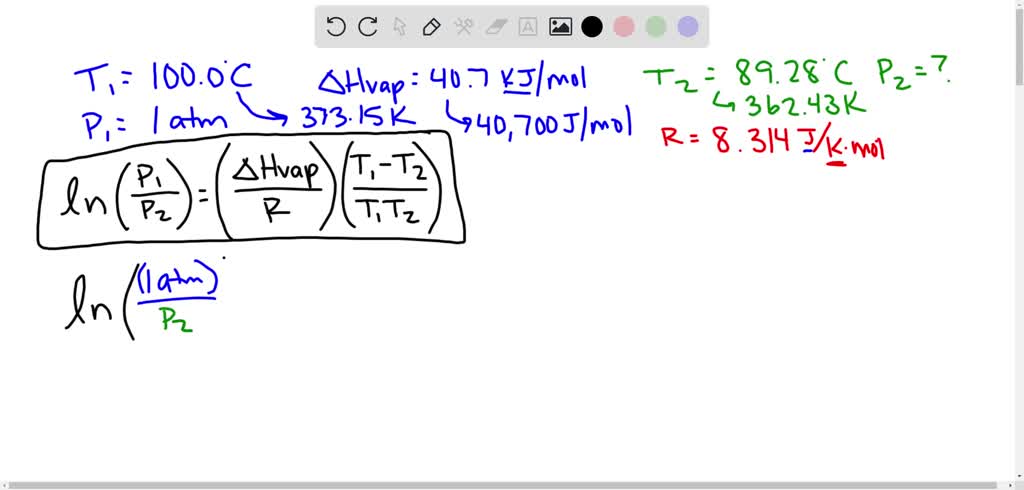
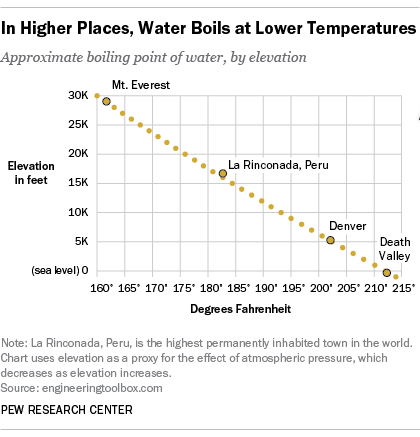
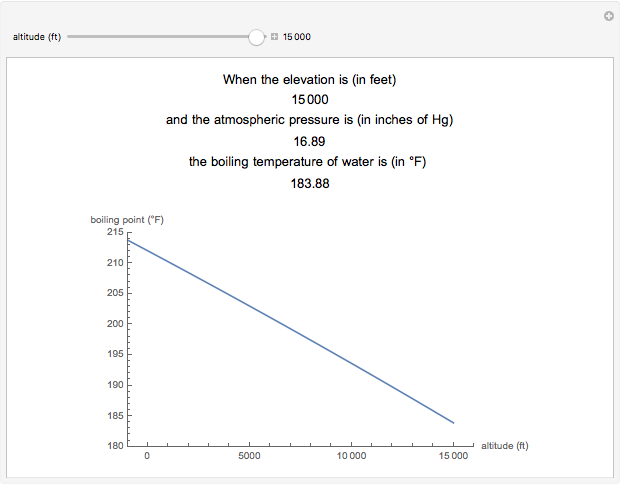
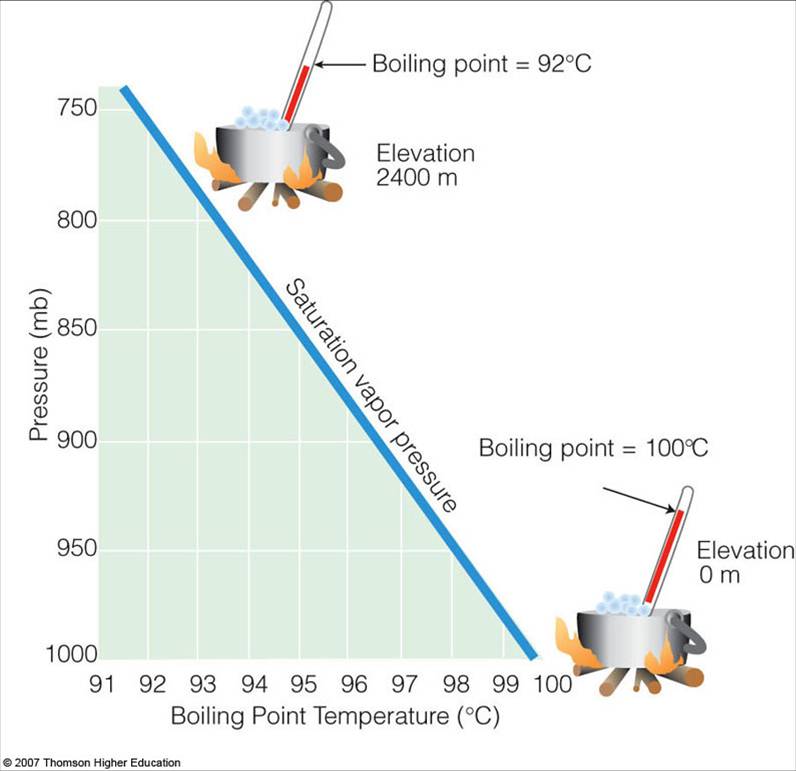
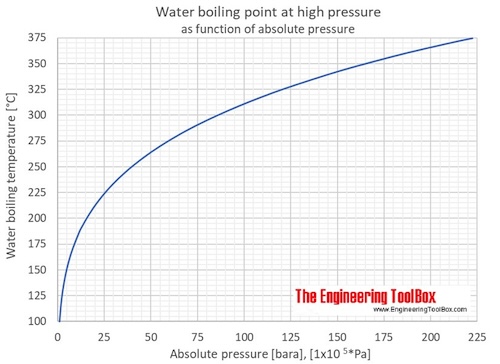
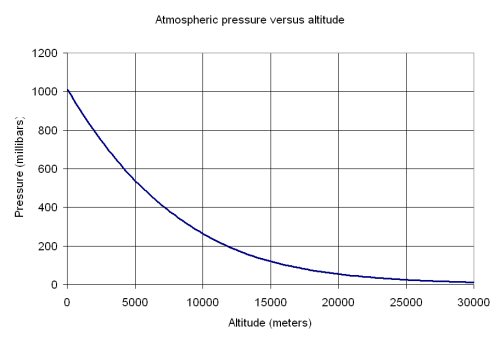
If water boils at 100 degrees C at sea level how high must one be for it to boil at zero degrees C? - Quora

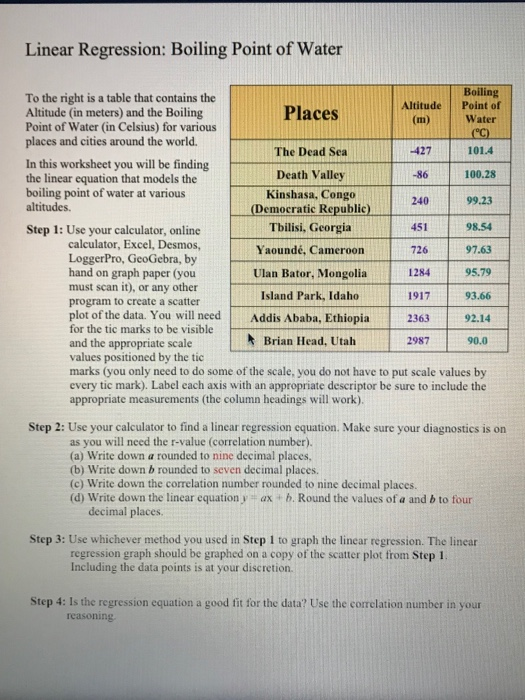
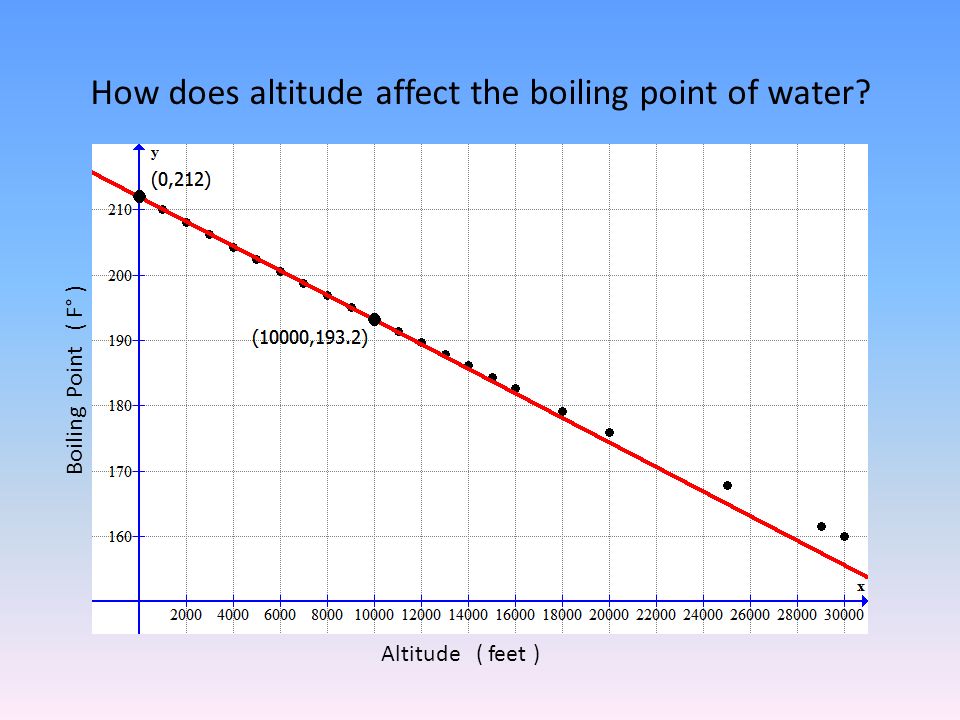
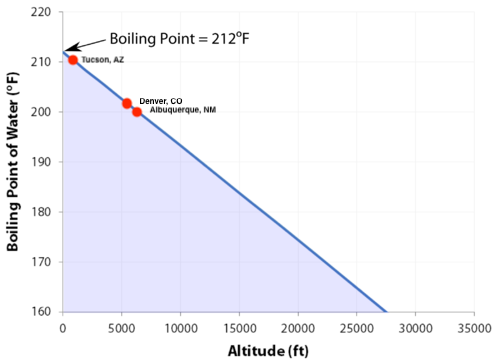
SOLVED:Boiling Point The relationship between altitude and the boiling point of water is linear. At an altitude of 8000 ft, water boils at 197.6^∘ F. At an altitude of 4500 ft, water

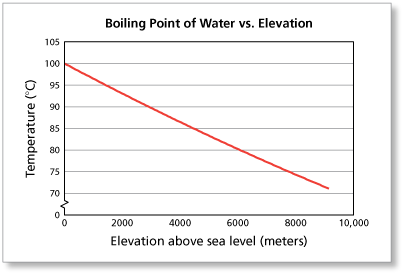
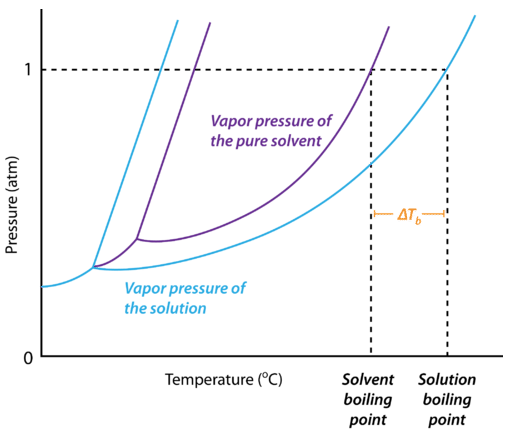
Boiling Point Overview & Calculation | How to Calculate Boiling Point - Video & Lesson Transcript | Study.com

Boiling Point Overview & Calculation | How to Calculate Boiling Point - Video & Lesson Transcript | Study.com