Titration Calculations Revision. titration - accurate neutralisation of an acid with an alkali data obtained can be used to do calculations equation used. - ppt download

If enthalpy of neutralization of HCl by NaOH is - 57kJmol^-1 and with NH4OH is - 50kJmol^-1 , then calculate the enthalpy of ionisation of NH4OH(aq) .

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa



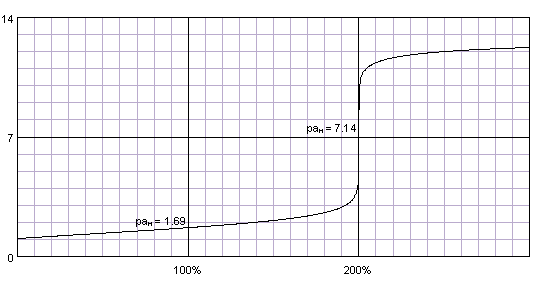

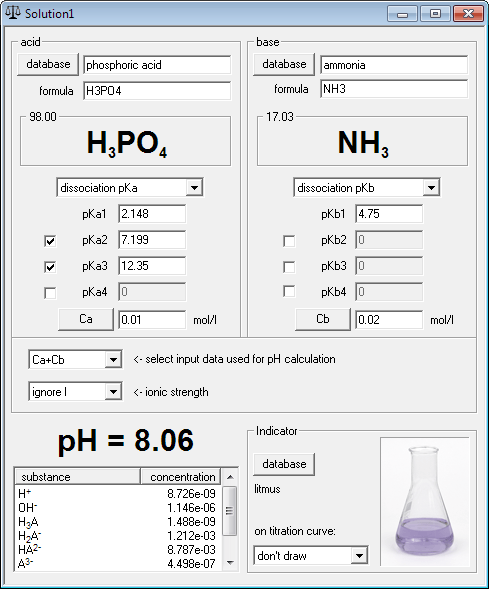




![4.4] Heat of neutralisation - Calculation - YouTube 4.4] Heat of neutralisation - Calculation - YouTube](https://i.ytimg.com/vi/x_993x_cBh8/hqdefault.jpg)